Listing ID #5741762
Company Information
Ask for more detail from the seller
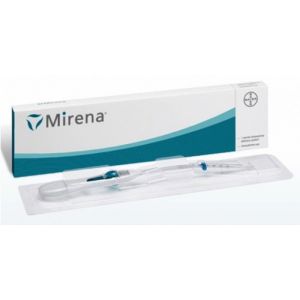
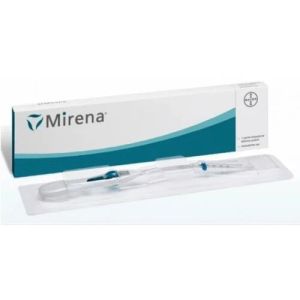
Contact Supplierintrauterine device - long lasting contraceptive for 10 years size and accessories: additional accessories like hysterometer / probe and loading / folding devices are provided on specific customer request. horizontal arm length in mm vertical stem lenght in mm approx. sound measuring range purity of copper as cu total active copper surface area 32 36 6-9 cm 99.99% 380mm the above information is indicative only. the final decision of selection of the correct model for the patient is to be made by the gynecologist/physician. desciption: each smb tcu 380a t frame is made from compound of low density polyethylene and barium sulphate (15% - 24%) wound with 176mg of 0.25mm diameter copper wire, and two copper sleeves on horizontal arm providing a surface area of 380mm 23mm . the 't' is equipped with high-density polyethylene filament for easy removal. it is packed together with an insertion tube and solid rod in a tyvek-mylar film pouch or mylar-mylar film pouch. centimeter scale printed in blue is provided on the insertion tube for exact positioning of the flange on the insertion tube before lnsertion. smb corporation has a new form of winding system for smb tcu 380a. this double copper winding system has an advantage over the single winding system. the double winding form rules out the possibility of protrusion and this system does not allow overlapping of wire. it is 99% effective intrauterine contraceptive device and does not affect female hormonal system. the smb iud's have under gone clinical studies in vietnam for 60 months and confirmed to be a safe and effective contraceptive. advantage of copper winding: winding is difficult to unwind smooth and uniform winding over the stem no protrusion or loose end near the stem hole uniform wire tension throughout the winding length makes it more elegant approved by meb netherlands health canada proven safe reversible long intrauterine life highly convenient requires no daily attention no touch technique is employed while insertion can be used by women who cannot use steroid contraceptives safe after abortion or miscarriage with no infection safe in hiv infected women on antiretroviral therapy and clinically well easy to load hormone free immediate effective safe contraceptive for breast feeding women conception is possible after removal of the device safe for all women including adolescents and women over 40 safe in women having no history of pelvic inflammatory disease(pid)