Listing ID #3560414
Company Information
Ask for more detail from the seller
Contact SupplierWe are sprawling over a large area with exceptional warehouse, which is well-equipped with cutting-edge technology and up-to-date management systems. With automated warehouse systems, we efficiently serve the bulk requirements. We believe in providing quality-approved products to the clients. For this, we make sure that all the inputs are procured from certified vendors after complete analysis of the quality.
| Test Parameter | Specifications |
|---|---|
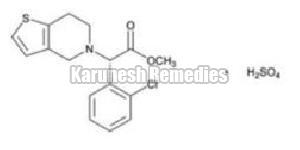
| Description | White to off-white powder |
| Solubility | Freely soluble in any acid, practically insoluble in water. |
| Identification | |
| a) By IR | The infra-red absorbance spectrum obtained from the sample should be concordant with spectrum obtained with that Clopidogrel bisulfate RS/WS. |
| b) By HPLC | The retention time of the major peak of the sample solution corresponds to that of the standard solution, as obtained in the assay. |
| c) Sulphate test | Should meet test for Sulfate |
| Loss on drying (%w/w) | Not more than 0.5 |
| Residue on ignition (%w/w) | Not more than 0.1 |
| Assay by HPLC (% on dried basis) | Not less than 97.0 and not more than 101.5 |
| Related substances by HPLC (%) | |
| Clopidogrel Related Compound A | Not more than 0.2 |
| Clopidogrel Related Compound B | Not more than 0.3 |
| Individual impurity | Not more than 0.10 |
| Total impurities | Not more than 0.5 |
| Clopidogrel Related Compound C | Not more than 0.5 |
| Residual Solvents by GC (ppm) | |
| Methanol | Not more than 3000 |
| Acetone | Not more than 5000 |
| Isopropyl Alcohol | Not more than 5000 |
| Methylenedichloride | Not more than 600 |
| n-Hexane | Not more than 290 |
| Ethyl acetate | Not more than 5000 |
| Toluene | Not more than 890 |